 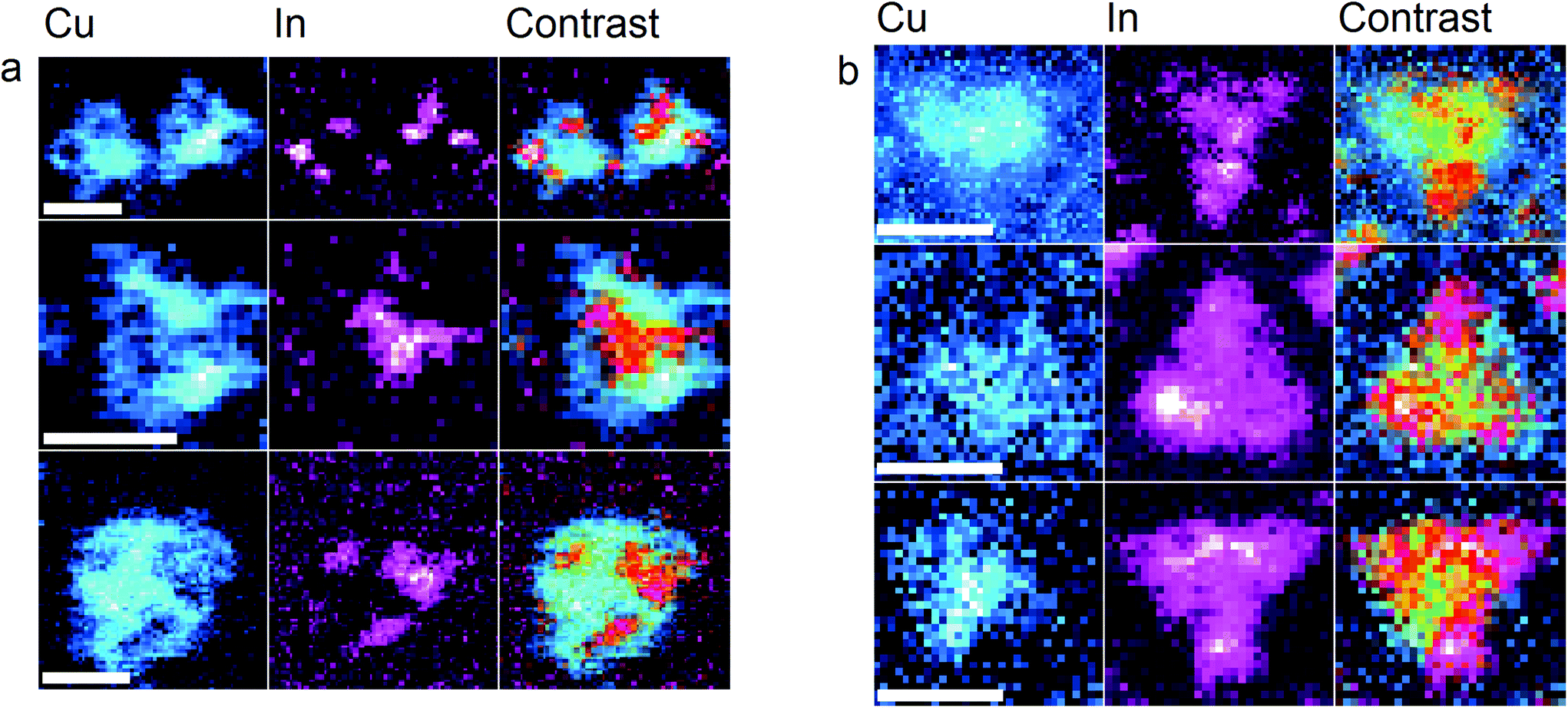
They have an increased energy density (1274 Wh kg −1) compared with high-temperature Na–S batteries 12, because Na 2S instead of Na polysulfides is the final discharge product 13, 14: Room-temperature Na–S batteries have been reported since 2006 11. As a result, great efforts have been devoted to lower the working temperature and develop room-temperature Na–S batteries with enhanced safety. Such high temperature not only increases the cost of operation and maintenance, but also brings serious safety hazard due to the highly active molten electrodes, which directly restraints the widespread applications of high-temperature Na–S batteries 5, 8. However, the operating temperature of this system is generally as high as 300–350 ☌ to ensure a sufficient conductivity of sodium β-alumina solid-electrolyte and keep the polysulfides in a molten state, far exceeding the melting points of Na (98 ☌) and sulfur (115 ☌) 10. This rechargeable battery system has significant advantages of high theoretical energy density (760 Wh kg −1, based on the total mass of sulfur and Na), high efficiency (~100%), excellent cycling life and low cost of electrode materials, which make it an ideal choice for stationary energy storage 8, 9. The as-developed sodium–sulfur batteries deliver high capacity and long cycling stability. Indium triiodide as redox mediator simultaneously increases the kinetic transformation of sodium sulfide on the cathode and forms a passivating indium layer on the anode to prevent it from polysulfide corrosion. As verified by first-principle calculation and experimental characterization, the fluoroethylene carbonate solvent and high salt concentration not only dramatically reduce the solubility of sodium polysulfides, but also construct a robust solid-electrolyte interface on the sodium anode upon cycling. Herein, we report a room-temperature sodium–sulfur battery with high electrochemical performances and enhanced safety by employing a “cocktail optimized” electrolyte system, containing propylene carbonate and fluoroethylene carbonate as co-solvents, highly concentrated sodium salt, and indium triiodide as an additive. 
However, the safety concerns greatly inhibit their widespread adoption. High-temperature sodium–sulfur batteries operating at 300–350 ☌ have been commercially applied for large-scale energy storage and conversion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
